Effect of Confinement on Structure, Water Solubility, and Water Transport in Nafion Thin Films
Citation
Eastman, S.A.; Yager, K.G.; Kim, S.; Page, K.A.; Rowe, B.W.; Kang, S.; Soles, C.L. "Effect of Confinement on Structure, Water Solubility, and Water Transport in Nafion Thin Films"
Macromolecules 2012,
45 (19) 7920–7930.
doi: 10.1021/ma301289vSummary
We explore ordering of nafion at interfaces.
Abstract
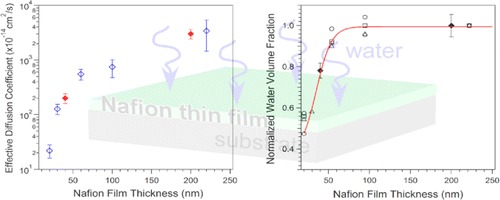
Fuel cells based on polymer electrolyte membranes (PEM) show promise as a means of energy conversion for a wide range of applications both in the transportation sector and for stationary power production due to their high charge density and low operating temperatures. While the structure and transport of bulk PEMs for fuel cell applications have been studied extensively, much less is known about these materials at interfaces and under confinement, conditions that are highly relevant in the membrane electrode assembly of a working PEM fuel cell. Using X-ray reflectivity, neutron reflectivity, grazing-incidence small-angle X-ray scattering, quartz crystal microbalance, and polarization-modulation infrared reflection–absorption spectroscopy, we have studied the structure, swelling, water solubility, and water transport kinetics as a function of relative humidity for confined polyelectrolyte films thinner than 222 nm. While the humidity-dependent equilibrium swelling ratio, volumetric water fraction, and effective diffusivity are relatively constant for films thicker than ca. 60 nm, we observe measurable suppressions of these properties in films less than ca. 60 nm. These effects occur at length scales that are relevant to transport (ion and water) in the polyelectrolyte binders found in the catalyst layer of the membrane–electrode assembly (MEA) of a functional fuel cell. The thin film methodology and findings presented here provide a platform to quantify and validate models of interfacial impedance used within the fuel cell community and have the potential to lead to improvements in MEA materials, design, and optimization.

