Mesoporous Carbon–Vanadium Oxide Films by Resol-Assisted, Triblock Copolymer-Templated Cooperative Self-Assembly
Citation
Bhaway, S.M.; Kisslinger, K.; Zhang, L.; Yager, K.G.; Schmitt, A.L.; Mahanthappa, M.K.; Karim, A.; Vogt, B.D. "Mesoporous Carbon–Vanadium Oxide Films by Resol-Assisted, Triblock Copolymer-Templated Cooperative Self-Assembly"
ACS Applied Materials & Interfaces 2014,
6 19288–19298.
doi: 10.1021/am505307tSummary
We describe conditions that enable the formation of vanadium oxide nanostructures using a triblock-copolymer organic template.
Abstract
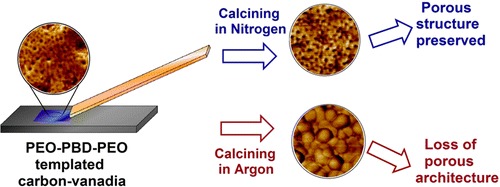
Unlike other crystalline metal oxides amenable to templating by the combined assemblies of soft and hard chemistries (CASH) method, vanadium oxide nanostructures templated by poly(ethylene oxide-b-1,4-butadiene-b-ethylene oxide) (OBO) triblock copolymers are not preserved upon high temperature calcination in argon. Triconstituent cooperative assembly of a phenolic resin oligomer (resol) and an OBO triblock in a VOCl3 precursor solution enhances the carbon yield and can prevent breakout crystallization of the vanadia during calcination. However, the calcination environment significantly influences the observed mesoporous morphology in these composite thin films. Use of an argon atmosphere in this processing protocol leads to nearly complete loss of carbon–vanadium oxide thin film mesostructure, due to carbothermal reduction of vanadium oxide. This reduction mechanism also explains why the CASH method is not more generally successful for the fabrication of ordered mesoporous vanadia. Carbonization under a nitrogen atmosphere at temperatures up to 800 °C instead enables formation of a block copolymer-templated mesoporous structure, which apparently stems from the formation of a minor fraction of a stabilizing vanadium oxynitride. Thus, judicious selection of the inert gas for template removal is critical for the synthesis of well-defined, mesoporous vanadia–carbon composite films. This resol-assisted assembly method may generally apply to the fabrication of other mesoporous materials, wherein inorganic framework crystallization is problematic due to kinetically competitive carbothermal reduction processes.

